By XH-Alan | 06 May 2026 | 127 Views
Sodium-Ion vs LFP: Can Sodium Batteries Challenge LFP’s Dominance in 2026?
Sodium-ion batteries have moved far beyond theoretical promise to become a tangible industrial force. Once dismissed as a niche complementary technology, sodium-ion is now entering mass commercial deployment, with manufacturers scaling production for energy storage and entry-level EVs. This raises a critical industry question: can sodium-ion batteries disrupt lithium iron phosphate (LFP), the current dominant chemistry for cost-sensitive applications?
1. Sodium-Ion’s Rapid Industrialization Journey
Sodium-ion technology has advanced at an unprecedented pace over the past two years. Today’s commercial cells exceed 300Ah in capacity, reach 175 Wh/kg energy density, and deliver cycle lives beyond 15,000 cycles. These metrics have transformed sodium-ion from a laboratory curiosity into a practical alternative for multiple market segments. Crucially, sodium-ion is not a single chemistry but a flexible platform whose performance can be tailored to specific applications through material and system design choices.
2. Core Cathode Technologies: Three Distinct Paths
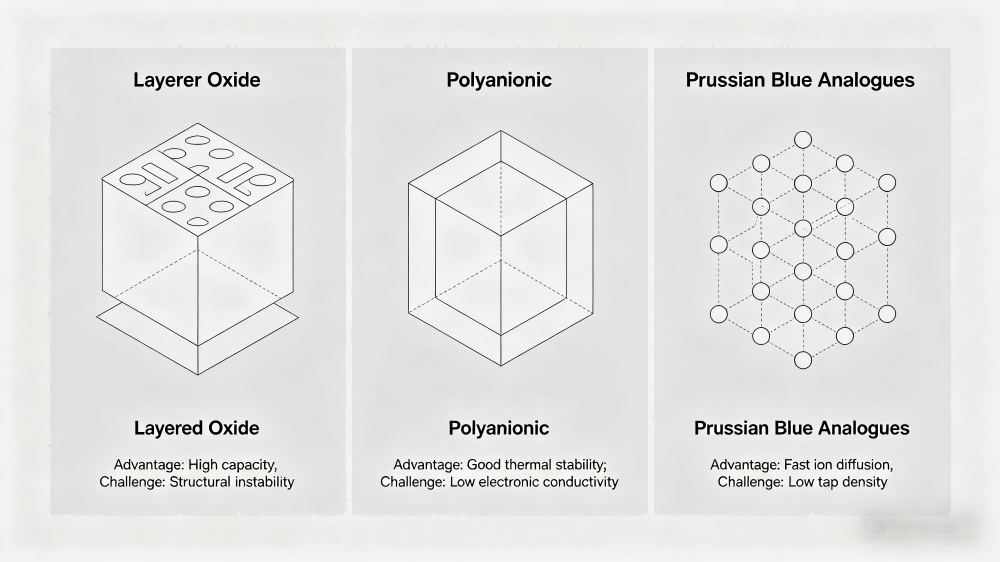
Sodium-ion performance varies dramatically based on cathode material, with three main technologies competing for market share:
- Layered Oxides: Optimized for energy density, similar to lithium NMC systems. They offer the highest voltage and capacity but face challenges with structural stability and thermal degradation.
- Polyanionic Materials: Prioritize safety and longevity, making them ideal for long-duration energy storage. They deliver exceptional cycle life and thermal stability but have lower energy density.
- Prussian Blue Analogues (PBA): Excel in cost and low-temperature performance. Their open framework structure enables fast ion diffusion, but they require careful manufacturing to address moisture sensitivity and gas generation issues.
3. Anode Innovation: Hard Carbon Leads the Way
Hard carbon remains the dominant anode material for sodium-ion batteries, offering a balance of capacity (250–350 mAh/g) and manufacturability. The primary challenges are low initial coulombic efficiency and the need for pre-sodiation to compensate for irreversible sodium loss during the first cycle. While alloy anodes offer higher theoretical capacity, their severe volume expansion issues have prevented commercial adoption to date.
4. Safety & Performance Head-to-Head: Sodium-Ion vs LFP
| Metric | Sodium-Ion | LFP |
|---|---|---|
| Energy Density | 100–175 Wh/kg | 160–210 Wh/kg |
| Cycle Life | 2,000–15,000+ | 3,000–10,000+ |
| Low-Temperature Performance | Excellent (-40°C) | Moderate |
| Resource Dependency | No lithium required | Requires lithium |
Safety profiles also differ by chemistry: polyanionic sodium-ion matches LFP’s thermal stability, while layered oxide systems behave more like high-energy lithium batteries. PBA systems have low thermal runaway risk but require mitigation for potential toxic gas release.
5. The Verdict: Coexistence, Not Replacement
Sodium-ion batteries will not fully replace LFP in the near term. Instead, the market is moving toward a multi-chemistry future where each technology is deployed according to its strengths. Sodium-ion will dominate low-temperature applications, grid energy storage, and entry-level EVs, while LFP will maintain its lead in mainstream EVs and high-energy storage systems. This complementary approach will drive down overall battery costs and accelerate the global energy transition.
AA vs AAA Batteries: Complete 2026 Guide – Differences, Sizes & Which Lasts Longer
How to Choose the Perfect Lithium-Ion Battery Cell: Expert Selection Guide 2026
Leave a Reply
Your email address will not be published.Required fields are marked. *
POPULAR BLOG
CATEGORIES
